Want to write a piece for 3DHEALS Expert Corner? Email us: info@3dheals.com
Where do you see yourself in 5 to 10 years? It’s a common question asked during the interview process. The purpose of the question isn’t to obtain some clear objective answer. Rather, the purpose of the question is to assess one’s ambitions and to determine if the candidate is a career goal-oriented. Let me augment the question slightly. Where do you see your 3D printing program in 5 to 10 years?
The question may have posed a real challenge to early adopters of “point-of-care” manufacturing several years ago. However, we are now in the early stages of seeing real reimbursement for hospital-based 3D printing programs. The Category III CPT code 0559T and its modifier 0560T can be used when producing a 3D printed patient-specific model to aid in pre-operational planning. While the Category III CPT code 0561T and its modifier 0562T pertain to 3D printed patient-specific surgical guides. The implementation of these codes (as of July 2019) is a real milestone within the point-of-care manufacturing community, but there’s still much work to be done.

Photo Credit: Robert Wesley
For starters, Category III CPT codes are used to track the utilization of emerging technologies, services and procedures. The Category III CPT codes description does not establish a service or procedure as safe, effective or applicable to the clinical practice of medicine. While some private health insurance companies may reimburse for some portion of a 3D printing service, don’t count on Medicaid or Medicare to until the Category I CPT codes exist. A private health insurer can deny the claim of a 3D printed model for patient care on the basis that 3D printed models have not been proven effective in significantly improving the course of patient care and will be deemed not medically necessary.
So, how does the community tackle this challenge?
The Radiological Society of North America (RSNA) and the American College of Radiology (ACR) plan to initiate a new medical 3D printing clinical data registry to collect 3D printing data at the point of clinical care. A joint ACR-RSNA committee will oversee the registry intended to pilot in the fall of 2019. The ACR’s National Radiology Data Registry (NRDR) system will host the 3D printing registry. The registry will provide the quantifiable data supporting the claim that 3D printing to create anatomic models and guides has a significant positive impact on patient care such as reducing blood loss, time in the operating theatre, length of postoperative stay, readmissions; minimizing complications, increasing patient consent, and other important metrics that have long been difficult to capture and report in peer-reviewed literature.
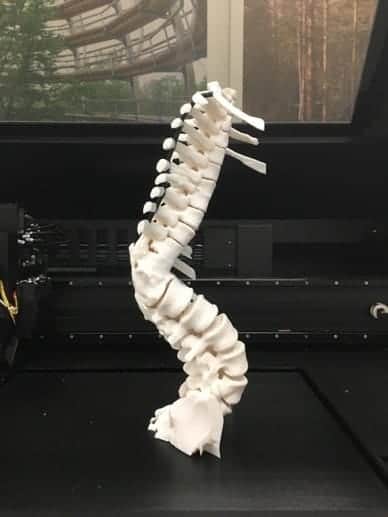
On an individual level, say the tech, engineer, or physician running the day-to-day operations of the 3D printing program, more challenges appear. When in the process of patient imaging to 3D printing are these codes implemented? It’s becoming the popular opinion that one (or more) of these codes is charged under radiology, which makes sense, but only if, at the time of ordering the imaging (CT, MRI, or 3D ultrasound) or interpreting the images, the physician knows an anatomic model will be 3D printed. At the same time, imaging standards for 3D printing typically fit for virtual 3D reconstructions, for which there are already Centers for Medicare & Medicaid Services (CMS) reimbursement through codes 76376 and 76377. It’s important to note that 3D recon (or post-processing) codes cannot be used in conjunction with the new 3D printing codes. This clash may stem from that a virtual 3D reconstruction is one of the steps in producing an anatomic 3D printed model.
Others are under the opinion that the charge or reimbursement should reroute to the parent department of the 3D printing program. After all, not all 3D printing programs are birthed from radiology, rather some programs origins are in plastic, orthopedic, or cardiothoracic surgery. Or does the charge occur at the end-user, the operating room? One goal of these codes is to obtain some sort of standardization across hospital-based 3D printing programs. If that is the case, then having a unified answer to this question plays an important role.
Placing the code under a radiology charge may be more feasible when referencing the Radiological Society of North America (RSNA) 3D printing Special Interest Group (SIG): guidelines for medical 3D printing and appropriateness for clinical scenarios. Published in 2018, this article from the journal 3D Printing in Medicine provides a list of many diagnoses categorized in a scoring system (1 through 9) based on the frequency of anatomic 3D printing associated with the said diagnosis. The higher the rating, the more likely an anatomic 3D printed model is considered. The rating and diagnosis can only be taken into consideration as the surgeon will have the final say. Not every Truncus Arteriosus (rating 9) will be 3D printed, but not every Ventricular Septal Defect (rating 3 to 5 depending on size and complexity) is automatically ruled out.
In the case of an audit, or perhaps for the sake of traceability, integrating a 3D printing program’s workflow with the institution’s electronic health record. Take Epic for example. Epic Systems is one of the largest providers of health information technology, used primarily by large U.S. hospitals and health systems to access, organize, store and share electronic medical records Hospitals that use this software held medical records of 64% of patients in the U.S. and 2.5% of patients worldwide. Creating custom orders in Epic, like the workflow for 3D printing, could take weeks to months at a time with no CPT code to tie the order to. However, now a hospital can create its own customizable 3D printing workflow based on its needs. As annoying as it can be at times, I prefer to gather as much information as I can upfront with multiple physician approvals from imaging to 3D print. Data input such as expected surgical date, priority, turnaround time, anatomy, diagnosis, imaging, and accession number, and sterility capability are worthwhile collecting. Further add-ons in the workflow can include scheduling time for physician review of segmentation and the 3D computer-aided design (CAD) prior to printing, with options to record the time and date of the approvals, when the model has started and finished printing, who the model was delivered to and when.
Where do you see your 3D printing program in 5 to 10 years? I have had to answer this question several times when developing proposals, not just for my program but for other hospitals as well. I would say the future is hopeful. More medical centers are dipping their toes in and adopting this technology every year, all of which will have data to contribute to the registry. In 2 years, we may very well have the quantifiable data needed to apply for Category I CPT codes for 3D printing anatomic models and surgical guides. Three years from now, the challenge of seeking reimbursement for the use of this technology and service may seem like a distant memory.
Ten years from now we may shift our focus to 3D printing metal, tissues/organs, and or pharmaceuticals in-house for patient care and how to get those services reimbursed.
About the Author:

Robert Wesley. 3D Printing Engineer, St. Louis Children’s Hospital, USA.
Bio: Robert is an experienced biomedical engineer, who has managed hospital-based 3D printing programs. He has acted as a consultant non-profit medical centers looking to invest in the 3D printing space in the Southwestern, Midwestern, and Southeastern regions of the United States. He works closely with research teams in designing and producing prototypes, and with clinical teams to identify anatomy, design and manufacture models, and apply quality standards to the entire process. Focused on capturing tangible cost-savings, he works with business development, finance and accounting, and with supply chain to track the utilization of these in-house centers versus outsourcing to external vendors.
Related Courses:
Introduction to 3D Printing in Hospitals by Robert Wesley
Related Articles:
3D Printing in Hospitals: Challenges and Solutions, a Children Hospital Perspective
Reimbursements for 3D Printed Anatomical Models and Surgical Guides – Decoding the CPT Codes
Pricing Strategies for 3D Printing Related Medical Devices
Healthcare 3D Printing Clinical Trials
Cardiac 3D Printing: The Heart of the Matter
3D Printing Has Come of Age But How Safe Are the Devices Going Into Our Mouth?
Idea to Implementation: Reimbursement, the Elephant in the Room




 Sep 28, 2019
Sep 28, 2019 





Comments